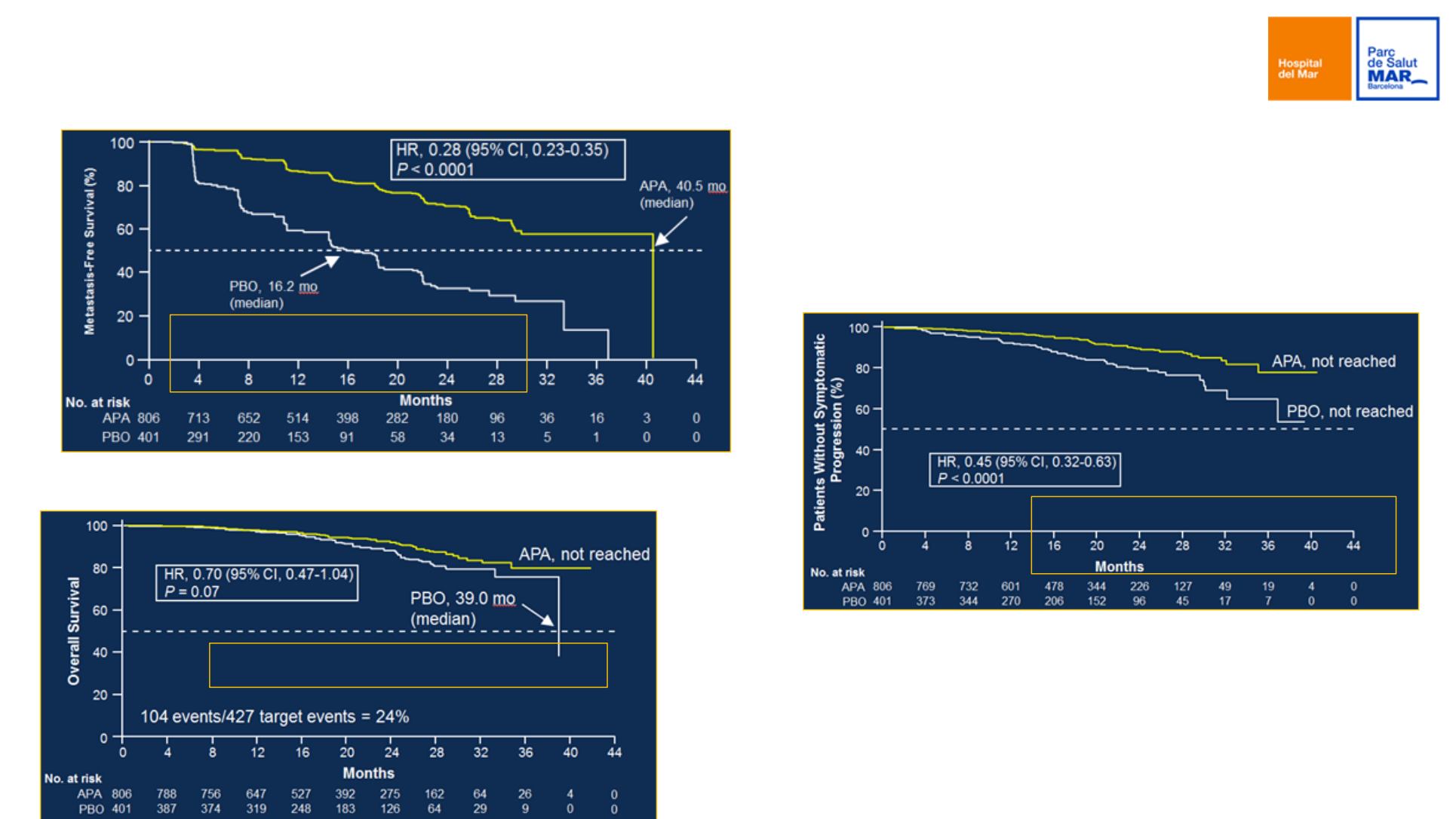
SPARTAN phase III trial
Primary endpoint: MFS
72% risk eduction of M1
progression or death
Secondary endpoint: OS
30% reduction risk of death
Secondary endpoint: time to symptomatic
progression
55% reduction risk of
symptomatic progression
Smith et al. Apalutamide Treatment and Metastasis-free Survival in Prostate Cancer. N
Engl J Med. 2018 Apr 12;378(15):1408-1418.








